3 Biomedical Industries That Don't Exist (But Should)
Disease treatment is the only paradigm that exists. While human capital deteriorates globally, three untapped industries sit at virtually zero investment and could reverse Idiocracy.
The global biomedical enterprise ($250 billion/year in pharma R&D, $47 billion/year from NIH, trillions in healthcare delivery) operates almost entirely within a single paradigm: disease treatment.
Fix things after they break.
The industry spends ~$250B/year across 480,000+ clinical trials.
Over 200 oncology drugs approved with 6,300+ more in the pipeline — meaning every new entrant is fighting thousands of competitors for the same patients, same trial sites, same FDA reviewers
After $300B+ on Alzheimer’s, only 2 drugs exist with marginal efficacy — the worst ROI in the history of pharma R&D
Each new drug costs $2.6B, takes 10–15 years, and has a 7.9% approval rate — meaning >90% of programs fail completely
Cancer 5-year survival improved ~20 percentage points in 50 years — 0.4pp/year and decelerating, despite exponentially increasing spend
Cost per QALY has risen from ~$50K in the 1990s to $100K–$500K+ today — each incremental gain costs more than the last
This paradigm is not failing — but it is mature, oversaturated, and structurally incapable of solving the upstream problem that causes 75% of all disease and death: biological aging.
And it has zero capacity to address the even larger opportunity beyond disease: making healthy humans better.
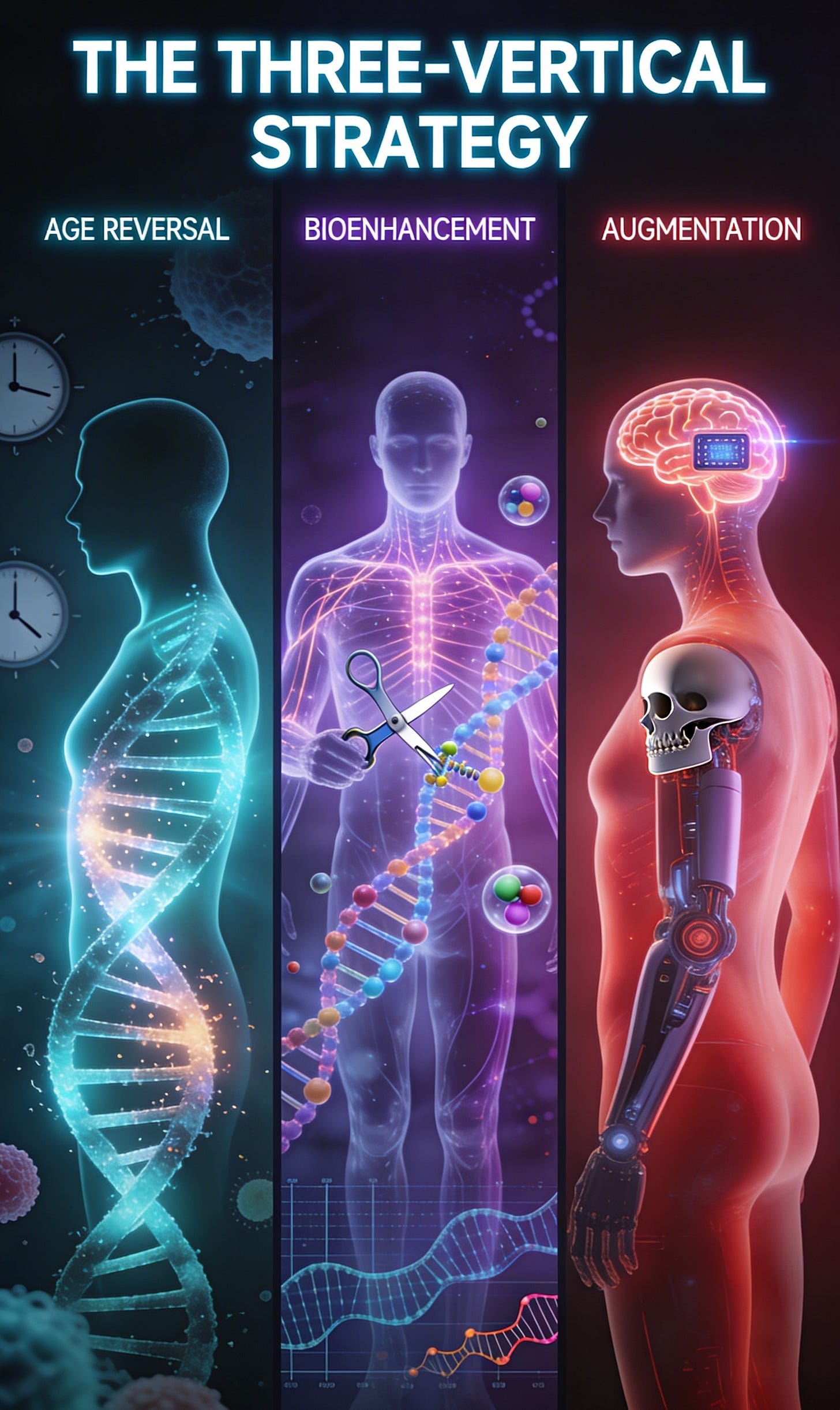
Three targets that should exist as biomedical industries but don’t:
Age reversal: Eliminating the root cause of most disease.
Bioenhancement: Making elite/healthy humans better at the molecular and genetic level.
Artificial augmentation: Boosting human capability through hardware, bionics, and brain-computer integration.
They don’t exist because Western regulatory, cultural, and institutional frameworks block them — not because the science is impossible or demand is absent.
Massively net-negative DEI-driven research burns funding on literature pollution instead of innovation.
Anti-meritocratic selection in academia filters for compliance over competence. Institutional groupthink protects status over progress.
Academic elites are enraged that a tech bro went “Founder Mode on Cancer” and had success; they’d rather you die if it helps them maintain social status and power (so they can virtue signal how irresponsible you are for trying to cure yourself).
Bloated regulations and predatory lawyers prevent companies from experimenting even on terminal patients.
Bioethicists who I’ve already argued are unethical have caused quantifiably insane amounts of harm (suffering and death) via philosophical grandstanding, moralizing, gatekeeping, and institutional influence. Because of them we guarantee deaths, prevent human experimentation in terminal patients, and we get zero valuable data.
The result: Stagnation and decay.
Disease treatment is one engine running at max capacity with diminishing returns (and even this could be massively improved by cutting regulations).
BUT AGE REVERSAL, BIOENHANCEMENT, AND ARTIFICIAL AUGMENTATION (TRANSHUMANISM) ARE 3 INDUSTRIES THAT HAVEN’T BEEN SERIOUSLY TRIED.
The first sovereign nation to build infrastructure across all three verticals won’t be competing with existing industries — it will be creating new ones that incumbents are ideologically and regulatorily prohibited from entering.
The TAM is potentially every human alive.
The competitive moat is a regulatory framework no other jurisdiction offers.
The timeline to first-mover advantage is ~5–10 years.
The cost of delay is measured in lives: 100,000 people die of age-related causes every day and the productivity boost from making elite humans “more elite” is potentially staggering.
Here I make the case that a sovereign wealth fund or national government should commit $50–100 billion over 10 years (Phase 1), scaling to $150–200 billion+ over 20 years.
I estimate that every dollar deployed will yield at least 100:1+ expected ROI and thus should be deployed as rapidly as infra can absorb it.
Part I: Biology Is Ready — Institutions Are Not
Engine 1: Age Reversal
Aging is the upstream cause of ~90% of cancer, 100% of Alzheimer’s, ~80% of cardiovascular mortality, and an estimated 75% of all deaths in developed nations.
~100,000 people die of age-related causes every day — 36.5 million/year.
The NIA spends ~$346M/year on aging biology — less than 1% of NIH’s budget.
All-time global investment in true age reversal is estimated at $15–20B. That’s what the US spends on healthcare in two days.
Only ~20–30 companies pursue epigenetic age reversal. Zero therapies approved. One human clinical trial exists (Life Biosciences, 2026).
We spend more annually on erectile dysfunction drugs ($5B+ market) than the NIA spends on the biology of aging.
The question “can biological aging be reversed?” was settled in 2024.
Cano Macip et al. (2024) — Gene therapy delivering inducible OSK factors to aged wild-type mice (~80+ human years equivalent) extended remaining lifespan by 109%. Frailty and organ deterioration reversed. Standard gene therapy vector, in principle translatable to humans.
Shift Bioscience (2025) — Single reprogramming factor (SB000) achieves cellular rejuvenation comparable to full OSKM without the cancer-inducing pluripotency. Directly addresses the primary safety concern.
Life Biosciences (2026) — FDA cleared the first human clinical trial of epigenetic reprogramming. Expected readout ~2028–2030.
Additional signals:
Klotho gene therapy (15–20% lifespan extension in mice), telomerase gene therapy (41.4% median extension)
Rejuvenate Bio’s three-gene cocktail simultaneously reversing obesity-diabetes-heart failure-kidney failure in mice, plasma dilution showing 2.61 years of biological age reduction in humans.
Five generations of reprogramming technology show clear safety convergence — from full OSKM (max rejuvenation, high cancer risk) through cyclic OSKM, OSK minus c-MYC, single-factor approaches, to emerging small-molecule reprogramming.
Each iteration reduces risk while preserving efficacy.
The path to safe, controllable reprogramming therapy requires investment and iteration, not fundamental breakthroughs.
Biology is not the bottleneck. Institutions are.
Engine 2: Bioenhancement (RNA Sandbox + Permanent Lock-Ins)
The optimal bioenhancement intervention in 2026 is programmable biology:
siRNA (silence any gene temporarily)
circRNA (express any protein for weeks without touching the genome)
base/prime editing (permanently install any point mutation with single-nucleotide precision)
These platforms are being developed for disease in Western clinical trials, but they’re target-agnostic — the LNP that delivers a PCSK9-silencing base editor for familial hypercholesterolemia could deliver that same editor to optimize cardiovascular risk in a healthy 30-year-old. Nobody does this because nobody is allowed to.
The technology is ready but the jurisdiction is missing. The FDA does not approve “enhancement” drugs and there’s no regulatory pathway to getting enhancers approved in any major jurisdiction.
Yet the unregulated nootropics market (mostly snake oil) is $5.5B (growing 15%+/year), the executive optimization grey market exceeds $10B, and DARPA spends an estimated $1–3B/year on classified enhancement R&D.
There is no biological law that says current human cognitive capacity or physical performance represents an optimum or peak — these are evolutionary tradeoffs under selection pressures that no longer apply.
Nobody knows the ceiling because nobody has tried to find it.
And fixation on “enhancement” drugs (which lead to bio tolerance) misses the smartest shortcut: temporary trial run (the sandbox) → lock in permanently if you like (and eventually reverse the permanent mods if you dislike them over time).
The “genetic sandbox then lock-in” strategy (the core value proposition):
Use reversible RNA interventions to test an enhancement in your own body, then commit to permanent gene editing whenever you want (i.e. if you are comfortable with the risk and/or there’s empirical validation, etc.).
The proof case exists — inclisiran (siRNA) silences PCSK9 with two injections/year for ~50% LDL-C reduction. A person could take it for 6–12 months, verify tolerance, then elect permanent PCSK9 base editing via Verve’s VERVE-102 (53–69% LDL-C reduction from a single infusion).
Someone else might opt for cognitive boost via BDNF Val66Met for activity-dependent BDNF (doesn’t treat a disease but may enhance learning).
Decide whether ongoing RNA treatments or permanent lock in. This pattern generalizes across every target where both RNA and editing approaches exist. And you will likely be able to even reverse the “permanent” edits in the future by re-editing back to homeostasis if you become dissatisfied.
Class A: Disease-related targets that enhance: PCSK9 knockout (88% CHD reduction), ANGPTL3 loss-of-function, APOE4→APOE3 conversion (60–90% Alzheimer’s risk reduction), myostatin silencing for muscle enhancement.
Class B: Targets that may enhance: COMT Val158Met, BDNF Val166Met, KIBRA T-allele, DRD2/DRD4 density, SLC6A4, FOXO3, SLC30A8 R325W, ACTN3, EPO pathway tuning, myostatin optimization, etc. (These are just preliminary ideas and not even close to what I have in mind for the full strategy.)
Delivery is cracking open fast.
Liver: Solved (GalNAc-LNPs).
Brain: Dyno’s AI-designed AAV capsids achieve 280-fold improvement over natural serotypes.
Muscle: advancing through DCA-conjugated siRNA and engineered AAVs.
New platforms (anellovectors, VLPs, synthetic exosomes): All in preclinical or early clinical development.
Enhancement operates across four modalities, each at a different readiness point:
Programmable Biology (highest EV, most ready) — The sandbox-to-lock-in framework deployed through an Enhancement Operating System (EOS). The EOS was designed by me and involves: eliminating genetic dampeners first, running permutational stack analysis on 50,000+ genomes at trait extremes, finding first-principles shortcuts (the GLP-1 lesson — complex traits have leverage points), installing supportive companion mods to offset trade-offs, using transient bridges, prioritizing asymmetric upside, stacking synergistically, and never stopping iteration as new variants, stacks, and delivery methods emerge.
AI-Designed Pharmacology (fastest revenue) — Nobody has designed an enhancement compound from first principles optimized for healthy humans. AI-driven molecular design explores chemical space orders of magnitude faster than traditional medicinal chemistry. Targets: selective dopaminergic optimization without addiction liability, orexin modulation for sleep efficiency, clean myostatin inhibitors, selective cortisol modulators. This generates revenue while the gene editing pipeline matures. Ideally you figure out zero-tolerance-pharmacology wherein drugs keep working at stable dose with minimal or zero drop-off in effect (likely less efficient than the RNA + gene mod lockins though).
Embryo Engineering (generational compounding) — Herasight’s CogPGT detects an average 8.5 IQ point difference between just 3 embryos, validated on 5,000+ sibling pairs. Orchid does whole-genome embryo sequencing (>99% of genome). MIT named embryo scoring a top 10 breakthrough of 2026. The progression: PGS on common variants → full WGS → permutational stack analysis → embryo gene editing → IVG expanding embryo pools to hundreds/thousands per cycle → generational compounding where each cohort’s gains become the next generation’s baseline. A sovereign program removes every constraint simultaneously — openly screen for positive traits, fund IVG, build the genomics engine, develop editing protocols, and track longitudinal outcomes at scale no private company can match. Eventually you could clone fleets of geniuses or other specialists if you wanted.
Delivery Infrastructure (enabling layer) — The single most leveraged investment. Every modality depends on getting cargo into the right cells. Owning or licensing next-generation delivery creates a chokepoint advantage: anyone who wants to do enhancement in any tissue must use platforms this program developed or controls.
Engine 3: Artificial Augmentation (Transhumanism)
When biological enhancement approaches its limits, the next frontier is bio-hardware integration — BCIs for cognitive augmentation beyond what genetics can deliver, engineered organs exceeding natural performance, bionic systems outperforming biological originals, synthetic biology creating capabilities with no natural equivalent.
$1B+ invested in BCIs alone in 2024–2025. Neuralink has implanted its N1 chip in five patients. Synchron demonstrated Apple Vision Pro control via brain signals. Precision Neuroscience received FDA clearance for its cortical interface. But all current BCIs are medical devices for disability — enhancement applications are 5–10 years from clinical reality.
This vertical is further out and technically harder.
Under dynamic allocation, it receives modest early funding that scales as technology matures. It adds optionality without diverting focus from the two core verticals, and biological enhancement clients are the natural first customers for augmentation technologies as they arrive.
What do I mean by dynamic allocation?
The program continuously shifts capital toward whatever is producing the best results.
If age reversal is generating 10× returns and augmentation isn’t ready yet, augmentation stays small.
If a BCI breakthrough suddenly shows exceptional ROI, capital flows there fast.
Nothing is sacred except the mission and the math.
Artificial augmentation starts with modest funding under this model — it’s further out and technically harder. But maintaining a presence keeps the option alive.
And the enhancement clients from Vertical 2 are the natural first customers for augmentation technologies as they arrive, so the demand pipeline is built-in.
Part II: Why Western Institutions Cannot Lead This
The FDA approves drugs to treat diseases.
Aging isn’t classified as a disease.
“Making healthy and smart people healthier and smarter” isn’t treating a disease.
No pathway exists for enhancement gene editing, positive-trait embryo screening, cognitive BCIs in healthy adults, or AI-designed enhancement compounds.
The entire apparatus (IND to Phase 3 to NDA) is built for disease treatment.
Estimated time to reform: 15–25 years, requiring Congressional legislation and political momentum (you probably won’t get the reform unless China creates a Prisoner’s Dilemma by going full throttle age reversal and enhancement… I’m hoping they do!)
Western bioethics treats enhancement and upgrades as inherently immoral. NIH study sections are biased against enhancement proposals. Academic careers punish researchers who pursue it and media coverage is overwhelmingly negative.
The entire pipeline — graduate school to faculty to NIH grant to clinical trial to FDA approval to insurance reimbursement — is optimized for woke status quo and “better disease management” and “healthspan” or whatever bullshit they’re pushing with insanely low ROI.
The US biomedical establishment will not lead this for the same reason Kodak didn’t lead digital photography: the institutional cost of transformation is too high for any individual actor to bear — it will break them mentally.
Part III: The Sovereign Opportunity
These verticals require patient capital (15–20 year horizons), regulatory authority (new approval pathways from scratch), scale ($50–100B+), risk tolerance, and geopolitical ambition.
The only capital class combining all five is sovereign wealth.
Why the Gulf States: Regulatory blank slate — no FDA legacy or woke political contamination.
Hevolution Foundation has committed $400M+ to aging biology.
Vision 2030 prioritizes diversification.
Existing medical tourism infrastructure.
Cultural alignment with civilizational-scale projects.
“We will save humanity from aging” is more compelling than another financial center — and would compete with nobody.
China is a potential competitive threat: $450B+/year R&D, regulatory flexibility, overrepresentation in aging biology talent. But lower international trust and geopolitical constraints create a positioning opportunity: the Gulf as the trusted, rigorous, internationally collaborative alternative to both Western paralysis and Chinese opacity. The window is 5–10 years.
Part IV: The Three-Vertical Strategy
Vertical 1 — Age Reversal ($40–80B over 15 years)
The civilizational mission.
Every year LEV is accelerated is worth $10–50T in reduced mortality and healthcare savings.
A 10× funding increase could compress timelines by 10–15 years — a 500:1 return.
Structure: National Age Reversal Research Institute (campus-scale), 50+ parallel delivery approaches, 200+ funded research programs, self-driving labs multiplying throughput 10–100×, dedicated clinical trial infrastructure, and aggressive talent recruitment (top 500 aging researchers globally at 3–5× market compensation with $5M–$50M unrestricted lab budgets).
Vertical 2 — Bioenhancement ($15–30B over 10 years)
The commercial engine, dynamically allocated across modalities based on evolving expected value:
Programmable Biology (~40–50%, $8–20B): EOS Genomics Engine ($1–2B) for 50,000+ genomes at trait extremes and permutational stack discovery. siRNA programs ($2–4B), circRNA platform ($2–4B), base/prime editing programs ($3–6B), target characterization and safety ($1–3B), and shared delivery R&D ($2–5B).
AI Pharmacology (~15–20%, $3–8B): AI drug design platform, de novo enhancement compounds, clinical trials in healthy adults, and elite performance clinic network (20+ locations, $100K–$500K/year programs). Revenue from year 2–3.
Embryo Engineering (~15%, $3–6B): Advanced polygenic screening center, IVG crash program, embryo editing R&D as somatic safety data matures, and longitudinal outcomes tracking.
Shared Infrastructure (~10%, $1.5–3B): Performance measurement lab, longitudinal enhancement database, regulatory standards body, and enhancement medicine training — creating a medical specialty that doesn’t exist.
The Revenue Flywheel: Sovereign authority creates legitimacy → 10,000 early clients at $100K/year = $1B/year by year 2–3 → revenue funds R&D → better results attract more clients → AI + sovereign regulatory flexibility compress timelines → published results create exponential demand → by year 5–7, enhancement revenue ($5B–$15B/year) exceeds R&D spend. The iPhone model: target elites initially (e.g. Bryan Johnson, LeBron, etc.), leverage profits to scale.
The endgame is pharma displacement. If you can permanently knock out PCSK9 with a single base edit, why take a statin daily for 40 years? If you can reverse epigenetic age and eliminate the upstream cause of most chronic disease, why would anyone need thousands of downstream drugs treating consequences of aging? Age reversal + bioenhancement makes the existing $1.4T pharmaceutical market increasingly obsolete. The methods developed for enhancement produce actual cures faster and cheaper than the disease-focused model because they address mechanisms rather than symptoms.
The long-term vision is on-demand biological customization: tune cognition, body composition, mood, sleep, social confidence (and whatever specifics you want within each subcategory) — different profiles for different contexts via transient RNA, with permanent lock-in when safety data supports it. Permanent is always the goal when the individual wants it and the data is there. Dependency on transient interventions creates supply chain vulnerability. Transient RNA is the testing phase, not the endpoint.
Vertical 3 — Artificial Augmentation ($5–15B over 15 years)
The longer horizon.
BCI Research Institute ($2–4B)
Organ Engineering Lab ($1–3B)
Bionics Center ($1–2B)
Synthetic Biology Platform ($500M–2B)
BCI-Biology Integration R&D ($500M–1B)
The same sovereign regulatory advantage enables enhancement BCIs in healthy adults — something no Western jurisdiction will approve for a decade. Enhancement clients from Vertical 2 are the natural first customers.
Cross-Vertical Synergies: Shared delivery platforms serve all three verticals. Safety data from each de-risks the others. Enhancement clients become augmentation customers. Age reversal provides moral legitimacy that benefits the enhancement brand. The IP portfolio across all three creates a moat no competitor can replicate.
Part V: The ROI Case
NEOM costs $500B+ and competes with Dubai. Index fund diversification competes with every institutional investor.
This program costs $50–100B and competes with nobody.
$100B buys ~38 drugs through FDA approval in a field with 6,300 competing oncology programs.
That same $100B buys foundational IP, infrastructure, and workforce for three entirely new global industries — 5–10× the total historical investment in age reversal, with increasing marginal returns.
It targets the elimination of the upstream cause of every disease that healthcare spending currently manages, saves 36.5 million lives per year, and compounds over centuries.
The value at stake: $180T/year in mortality cost, $10–15T/year in age-related healthcare, $1.4T/year pharmaceutical market ripe for displacement. At $930B+ in PIF AUM, $100B over 10 years is ~10% of assets — aggressive by normal standards, rational at 100:1+ expected returns.
The “absorptive capacity” objection is circular. The Manhattan Project went from a handful of physicists to 125,000 employees in 3 years. Operation Warp Speed deployed $18B in under a year. For crash programs with clear objectives, absorptive capacity expands to meet committed capital.
Part VI: Risk and Mitigation
Technical (moderate, declining):
Reprogramming may not translate from mice to humans (20–30%).
Cancer risk from early trials (15–25%).
Delivery technology may plateau (20–30%).
Address through diversification: multiple parallel approaches, tissue-specific sequencing, and the recognition that even conservative outcomes create massive markets.
Geopolitical (significant, manageable):
US restrictions on talent/tech transfer (15–30%) — mitigated by global recruitment, money, and experimental freedom.
China moving faster (25–35%) — countered by speed and trust advantage.
International backlash on “enhancement” framing — neutralized by leading publicly with age reversal or just not giving in to woke psychology (like is done for crime).
Reputational (highest priority): The framing: the FDA’s risk calculus is woke and unethical.
Every year of regulatory delay on a working therapy is 36.5 million deaths.
The program must be rigorous, transparent, and participant-protective but should not be afraid to pay anyone to participate in trials.
Deliver results, and criticism becomes what it always was: jealousy from those who are missing out.
Part VII: Implementation
Phase 1: Foundation (Years 1–3, $15–25B): Establish regulatory authority with approval pathways for all three verticals. Build campus-scale research institute, 5–10 self-driving labs, 10+ GMP gene therapy manufacturing facilities, AI compute cluster, elite performance clinic infrastructure. Recruit 200 senior researchers at 3–5× market. Launch 500 postdoctoral fellowships/year. Fund 200+ research programs (10× current global capacity). Launch EOS Genomics Engine immediately. Begin siRNA enhancement programs, AI pharmacology, embryo screening services, and BCI fundamental research.
Phase 2: Clinical Translation (Years 3–7, $25–40B): Phase 1 trials for organ-specific age reversal (eye, liver, skin, immune — simultaneously). First enhancement trials in healthy adults across siRNA, base editing, and AI-designed compounds. Elite clinic network generating revenue. IVG milestones. First-in-human cognitive BCI enhancement trials. Launch $5–10B longevity/enhancement startup incubator. Attract 50–100 international companies.
Phase 3: Commercialization (Years 7–15, $40–70B, partially self-funding): First approved age reversal therapies. Approved enhancement protocols across modalities. Medical tourism at scale. Enhancement revenue $5B+/year. Government/military contracts. Consumer-grade products. Embryo editing entering clinical use. By year 15, $30–60B+/year in revenue making continued investment self-sustaining.
Total: $80–135B gross, $50–95B net after revenue over 15 years.
Part VIII: Why Now?
Five trends converging:
Biological proof-of-concept is fresh (2024 mouse data, 2026 first human trial).
Enhancement tools crossed the clinical threshold simultaneously in 2024–2025 (base editing, prime editing, circRNA, AI-designed delivery).
AI is compressing timelines 2–10×.
Western institutions are paralyzed across every modality.
Public demand is exploding (embryo screening commercial, BCI investment >$1B, GLP-1 proving mass-market appetite for optimization).
Every year of delay costs ~36.5 million lives, $4.3T in US healthcare spending managing diseases rather than curing them, and competitive positioning that becomes exponentially harder to recover.
This failure creates the largest strategic vacancy in the history of biomedical investment. Three entire industries — age reversal, bioenhancement, and artificial augmentation — should exist but don’t.
What’s missing is a sovereign entity with the capital, regulatory authority, time horizon, and ambition to build what Western institutions won’t.
The competition: Nobody.
The cost of waiting: ~100,000 lives per day, a long-term Dark Age due to dysgenic momentum (fast-tracking Idiocracy), and a possible extinction event (as more low IQs gain power).